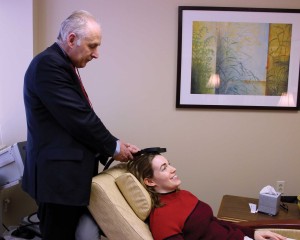
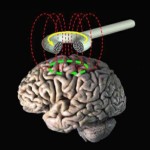
Transcranial Magnetic Stimulation (TMS) is a new brain stimulation technology that has been developed for the treatment of major depression. It has been FDA approved since 2008. I have personally used TMS for 18 years , initially on a research basis and clinically since 2008. TMS uses magnetic energy applied to the frontal part of the brain (called the prefrontal cortex, PFC) to re-energize circuits that are turned off by depression. After a course of TMS sessions the circuits come back on line and the symptoms of depression go away. TMS is a very safe treatment. The only common side effects are mild scalp discomfort or a transient headache. There is an extremely low risk of seizure. This is very rare.
 A TMS session last about 30 minutes. After the session you return to your normal activities right away including driving. A course of treatment with TMS is 36 sessions. Sessions are administered daily Monday to Friday for 6 weeks. There is then a three week taper phase of 6 sessions. If depression fully resolves many patients elect to have maintenance sessions of TMS to prevent depression returning. The frequency of maintenance sessions is typically 1-2 per month.
A TMS session last about 30 minutes. After the session you return to your normal activities right away including driving. A course of treatment with TMS is 36 sessions. Sessions are administered daily Monday to Friday for 6 weeks. There is then a three week taper phase of 6 sessions. If depression fully resolves many patients elect to have maintenance sessions of TMS to prevent depression returning. The frequency of maintenance sessions is typically 1-2 per month.
TMS has been shown to be effective in the treatment of depression, including treatment-resistant depression. It may help a range of other conditions: PTSD, OCD and auditory hallucinations in schizophrenia. Insurance coverage for TMS is variable. We will provide documentation of your TMS treatment at our center which you can submit to your insurance company to attempt to obtain reimbursement.
